Fibryga Reconstitution & Infusion Guide
Reconstitution guide1
The procedures below are provided as general guidelines for preparation and reconstitution of fibryga.
Part 1: Preparation
The fibryga package contains:
- 1 single-use bottle of fibryga concentrate
- 1 transfer device (Octajet)
- 1 particle filter (17-micron)
50 mL of sterile Water for Injection (sWFI) should also be used for reconstitution (not provided).
- Warm both the powder and sWFI in their closed bottles to room temperature. If a water bath is used, prevent water from coming into contact with rubber stoppers or caps of bottles. The temperature of the water bath should not exceed 37°C (98°F).
- Remove the caps from the fibryga and sWFI bottles to expose the central portion of the rubber stoppers. Clean the rubber stoppers with an alcohol swab and allow the rubber stoppers to dry .

- Peel away the lid of the outer package of the Octajet transfer device, leaving it in the clear outer package to maintain sterility.
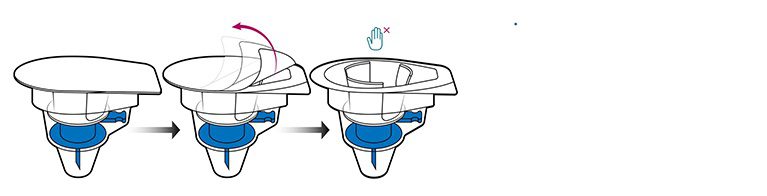
Part 2: Assembly
- Take the Octajet in its outer package and invert it over the fibryga bottle, pushing it down until the clips are locked. While holding onto the fibryga bottle, carefully remove the outer package from the Octajet. Be careful not to touch the blue water spike, and leave the Octajet attached firmly to the fibryga bottle.
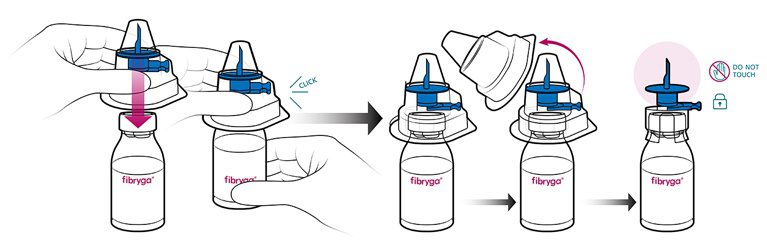
- With the fibryga bottle held firmly on a level surface, invert the sWFI bottle and place it at the center of the water spike. Push the blue plastic cannula of the Octajet firmly through the rubber stopper of the sWFI bottle.
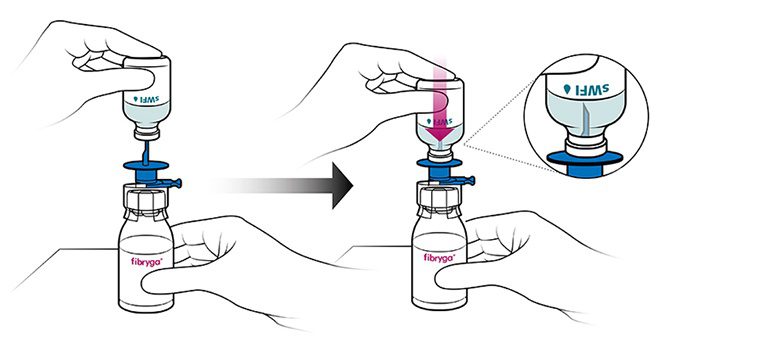
- Remove the distance ring and press the sWFI bottle down. sWFI will flow into the fibryga bottle.
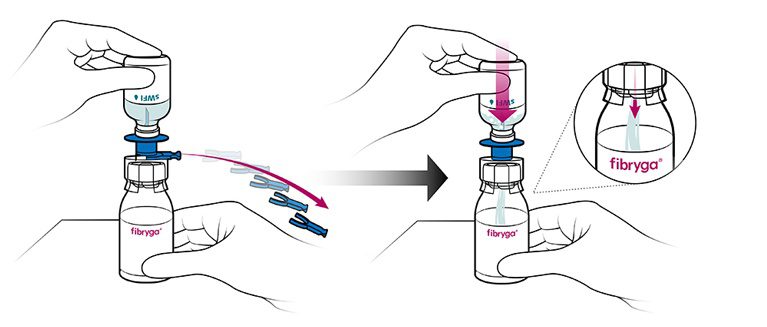
Part 3: Mixing and Transfer to Syringe
- When transfer of the sWFI is complete, gently swirl the fibryga bottle until the powder is fully dissolved. To avoid foam formation, do not shake the bottle. The powder should be dissolved completely within approximately 5 to 10 minutes.

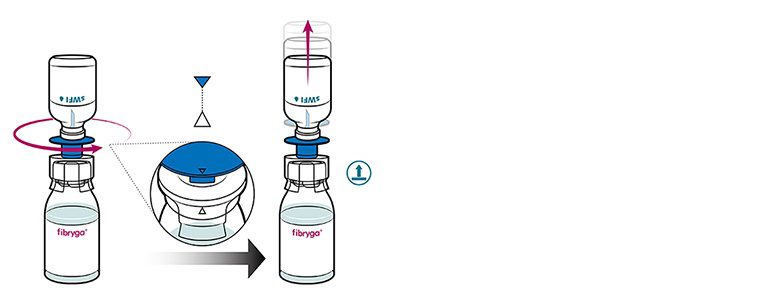
- Turn the blue sWFI bottle connector in either direction to bring the position markers together,
and remove the sWFI bottle together with the water spike. Keep the fibryga bottle upright to avoid leaking.
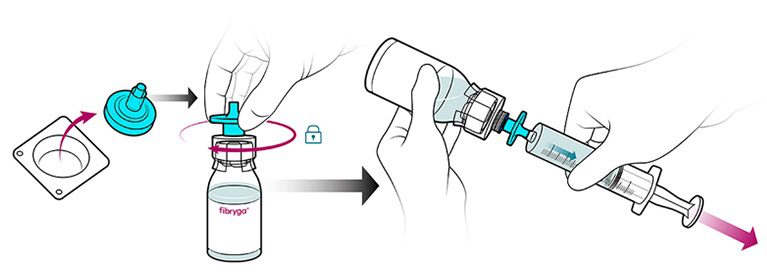
- Firmly connect the provided particle filter on the remaining Luer Lock on the fibryga bottle, and withdraw the solution through the particle filter into a syringe. Detach the filled syringe from the particle filter and discard the empty bottle and filter.
After reconstitution, the fibryga solution should be almost colorless and slightly opalescent. Inspect the reconstituted fibryga solution in the syringe for visible particulate matter and discoloration prior to administration. Do not use if particulate matter or discoloration are observed.
The powder should be reconstituted only directly before injection. After reconstitution, do not refrigerate or freeze the fibryga solution. Use the reconstituted fibryga solution immediately or within 4 hours after reconstitution. Discard any remaining fibryga solution.
Instructions for injecting fibryga1
For intravenous use only after reconstitution.
- Inspect the reconstituted fibryga solution in the syringe for visible particulate matter and discoloration prior to administration. Do not use if particulate matter or discoloration are observed.
- Do not administer fibryga in the same tubing or container as other medications.
- Clean the chosen injection site with an alcohol swab.
- Attach a standard infusion set to the syringe. Insert the needle of the infusion set into the chosen vein.
- Perform intravenous infusion. The rate of administration should be determined by the patient’s comfort level, at a recommended maximum rate of 5 mL per minute.
- After infusing fibryga, remove and properly discard the infusion set. After the infusion, remove the peel-off label containing the batch number from the fibryga bottle, and place it in the log book for record keeping. Discard the empty bottle.
References:
- Fibryga full Prescribing Information. Paramus, NJ: Octapharma; rev 2020.

